Noncovered hybrid stents are a significant advancement in Type A acute dissection repairs. They have potential for wider use due to simplicity. Safety and efficacy data remains limited with no guidelines. Standard use outside of aortic centers would need long term trial and registry data.
.jpg)
Hybrid Noncovered Open Stents for DeBakey Type 1 Acute Aortic Dissection
Repair: A Novel Approach
DeBakey type 1 acute aortic dissection (AAD) is a life threatening cardiovascular emergency caused by a tear in the internal lining of the aorta. This allows blood to flow between the layers of the aortic wall which can result in catastrophic complications such as aortic rupture or malperfusion. This condition necessitates emergency prompt surgical intervention and is considered one of the most challenging cases in cardiac surgical practice.
Traditional open surgical repair involves replacement of the ascending aorta with a tube graft. This is a shorter lifesaving operation but leaves a residual distal dissected aorta with long term complications. One innovative method to stabilize the residual aorta is the use of hybrid open (deployed directly in the aorta at surgery) noncovered stents.
Hybrid Noncovered Open Stents and the novelty of use
The only novel device presently available in this class is the AMDS (Ascyrus Medical Dissection Stent, Ascyrus Medical, Boca Raton, FL, USA). This was used first in Canada for positive remodelling of the residual aorta and gradually limited worldwide experience of this complex procedure has built up. Hybrid noncovered open stents (figure 1) stabilize dissected aortic wall from the inside allowing unhindered flow of blood in the true lumen. The stent is deployed in the open aorta at surgery under deep hypothermia and total circulatory arrest. Because they are uncovered, blood can flow through the stent to the head, neck and spinal branches of the aorta. This is crucial in preventing strokes and paraplegia. The self-expanding stent itself is made of a super alloy of Nickle and titanium (Nitinol) with a proximal Teflon felt cuff for stitching to aorta.
The FDA Breakthrough Device Program granted an exceptional approval in 2019 retaining the statutory standards for premarket approval, 510(k) clearance, and De Novo marketing authorization based on initial results of DARTS (Dissected Aorta Repair Through Stent Implantation) trial from Canada (1). FDA recognized the immense potential to improve mortality, morbidity and re-interventions of a complicated lifesaving emergency surgery. The device gained the CE (Conformité Européenne) mark for Europe and was approved by NICE, UK following a comprehensive review (IPG733, 03/08/2022).
The breakthrough advantages over existing techniques and technology
There was a slow but distinct recognition of the advantages compared to other covered open stents and endovascular stents used in TEVAR (Thoracic endovascular aortic repair) as limited worldwide experience started building up in Canada, Germany and UK.
Advantages of Hybrid Noncovered Open Stents
Clinical Outcomes
Previous experience with TEVAR and covered hybrid stents has shown positive remodelling of the downstream aorta is associated with better long-term outcomes, reduced re-intervention rates and better survival. Five publications including the DARTS trial have shown the technique to be safe and reproducible and significant benefits in false lumen obliteration, true lumen patency, improvement in malperfusion and positive remodelling of the residual diseased aorta. In the DARTS trial, AMDS promoted false lumen closure in 90% with significant reduction in DANE (distal anastomotic new entry). DANE is the single most important factor in 70% of the traditional repairs causing persistent false lumen patency and poor aortic remodelling with long term complications. Positive remodelling was observed in 77.1% in the proximal descending thoracic aorta, irrespective of false lumen status and true lumen remained stable or increased in 100% at 1 year. Even in a very high risk cohort in DARTS (malperfusion - 56.5%, preoperative stroke - 19.5%), postoperative stroke was 6.5% at 1 year with a complete resolution of false lumen aortic branches in all cases. The re-intervention rate in the largest series from Berlin was only 13% for persistent malperfusion after AMDS insertion (unrelated to stent).
Contemporary evidence for continued use is promising but based on limited short term data from trials and experience of individual aortic groups. Trial and registry safety data comprises of 30-day mortality, new neurological events, spinal cord ischemia, renal failure, SINE (stent induced new entry tears), device failure and device related re-interventions.
Is this the future of aortic dissection surgery?
AMDS is an adjunct procedure to conventional dissection repair in experienced hands. It does not entirely substitute for covered stents/ conventional surgery and has its own niche indications especially in patients who are very sick, with hemodynamic instability and malperfusion. There is an emerging expert consensus for concomitant stabilization of the downstream aorta at the time of the initial operation. AMDS would serve as an excellent tool for this in carefully selected patients at least in high volume aortic centers. The technique lends itself to training of non-aortic surgeons and does not increase the complexity of the repair so has potential for wider appeal and uptake.
Challenges and Considerations
Despite the potential benefits, hybrid noncovered open stents are not without challenges. The technique requires a high level of expertise in aortic dissection surgery and strategies for brain and visceral protection during prolonged periods of total circulatory arrest. Patient selection is critical, to exclude tears in the downstream aorta which is strict contraindication for use.
There are further challenges with consent and engagement of patients and their families given the emergency nature of surgery in very high risk patients. At this stage of development some oversight from patient care groups and compliance with local and national bypass for a new technology is advisable. Mentoring and proctorship is essential which in itself can be difficult due to emergency nature of these operations without adequate notice. Initial practice needs to be confined to surgeons experienced in high volume aortic centers only to select aortic surgeons preferably experienced in arch surgery and use of hybrid stent grafts. They can provide a landing zone for percutaneous devices however, there is very little experience of further open surgery in stented parts of the aorta and it is likely to be extremely challenging if the stents become incorporated in tissues or become infected. Further comparison to existing techniques and devices preferably in a randomized control setting or with Bayesian analysis is necessary to define the role of AMDS.
Conclusions
Open noncovered stent grafts represent a major technical advancement as an adjunct procedure for acute dissection repairs. It can be easily adopted for wider use by non-aortic surgeons due to simplicity of use and deployment. Initial safety and efficacy data has confirmed feasibility but long term trial and registry data is required to formulate recommendations for standard use.
Figures 1
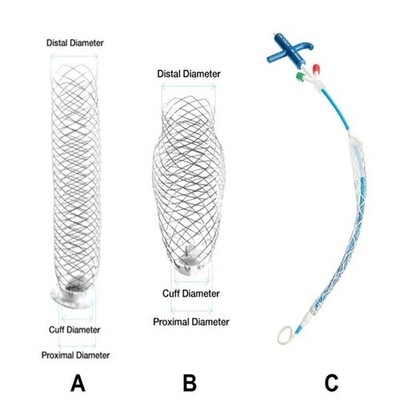
The AMDS device (A,B,C)
Figure 2
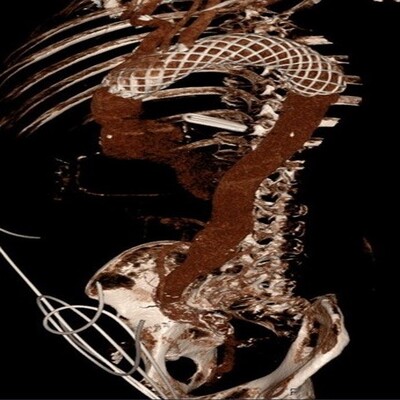
CT reconstruction of AMDS device in-situ in the thoracic aorta
References -